CRISPR Just Cured Sickle Cell Disease — Here’s the Biology Behind It
For the first time in history, a genetic disease is being permanently corrected inside living humans. How does it work, and what does it mean for medicine?
In December 2023, the U.S. Food and Drug Administration approved Casgevy — the world’s first CRISPR-based medicine cleared for human use. It treats sickle cell disease, a painful and life-shortening genetic disorder that affects roughly 100,000 Americans, most of whom are of African descent.
This is not just a medical milestone. It is the moment a technology once confined to laboratory petri dishes crossed into actual human bodies and rewrote the rules of what “treatment” means. To understand why this is so significant, we first need to understand the disease it targets — and the microscopic scissors that fix it.
What Is Sickle Cell Disease?
Your red blood cells are supposed to be round and flexible — like soft donuts that can squeeze through the narrowest capillaries in your body. In sickle cell disease, a single mutation in the HBB gene causes hemoglobin (the protein that carries oxygen) to behave abnormally under low-oxygen conditions.
Instead of staying dissolved and functional, mutated hemoglobin molecules clump together and form long, rigid rods. These rods distort the red blood cell into a crescent — a “sickle” shape. Sickled cells are stiff, sticky, and short-lived. They clog blood vessels, starve organs of oxygen, and cause episodes of excruciating pain called vaso-occlusive crises.
It takes just one wrong letter in 3 billion to sentence someone to a lifetime of pain. The mutation changes a single amino acid — glutamic acid becomes valine — and that tiny swap changes everything.
— Molecular basis of sickle cell pathologyBecause sickle cell disease is autosomal recessive, a person must inherit two copies of the mutated gene — one from each parent — to develop the full disease. Carriers (one copy) generally live normal lives. This inheritance pattern is key to understanding why CRISPR offers such a clean solution.
Enter CRISPR: Molecular Scissors That Read DNA
CRISPR-Cas9 stands for Clustered Regularly Interspaced Short Palindromic Repeats — a mouthful that describes a natural immune system bacteria evolved to fight viruses. Scientists discovered they could repurpose this system as a precise gene-editing tool.
Here is the simplest way to understand it: CRISPR is a two-part system consisting of a guide RNA (a short piece of RNA designed to find a specific DNA sequence) and the Cas9 protein (an enzyme that acts as scissors). The guide RNA leads Cas9 to an exact location in the genome, and Cas9 makes a cut.
Earlier tools like zinc finger nucleases and TALENs could also edit genes, but required months of custom protein engineering per target. CRISPR only requires designing a short RNA sequence — something that takes days and costs a fraction of the price. This accessibility transformed research and, ultimately, medicine.
How Casgevy Actually Works — Step by Step
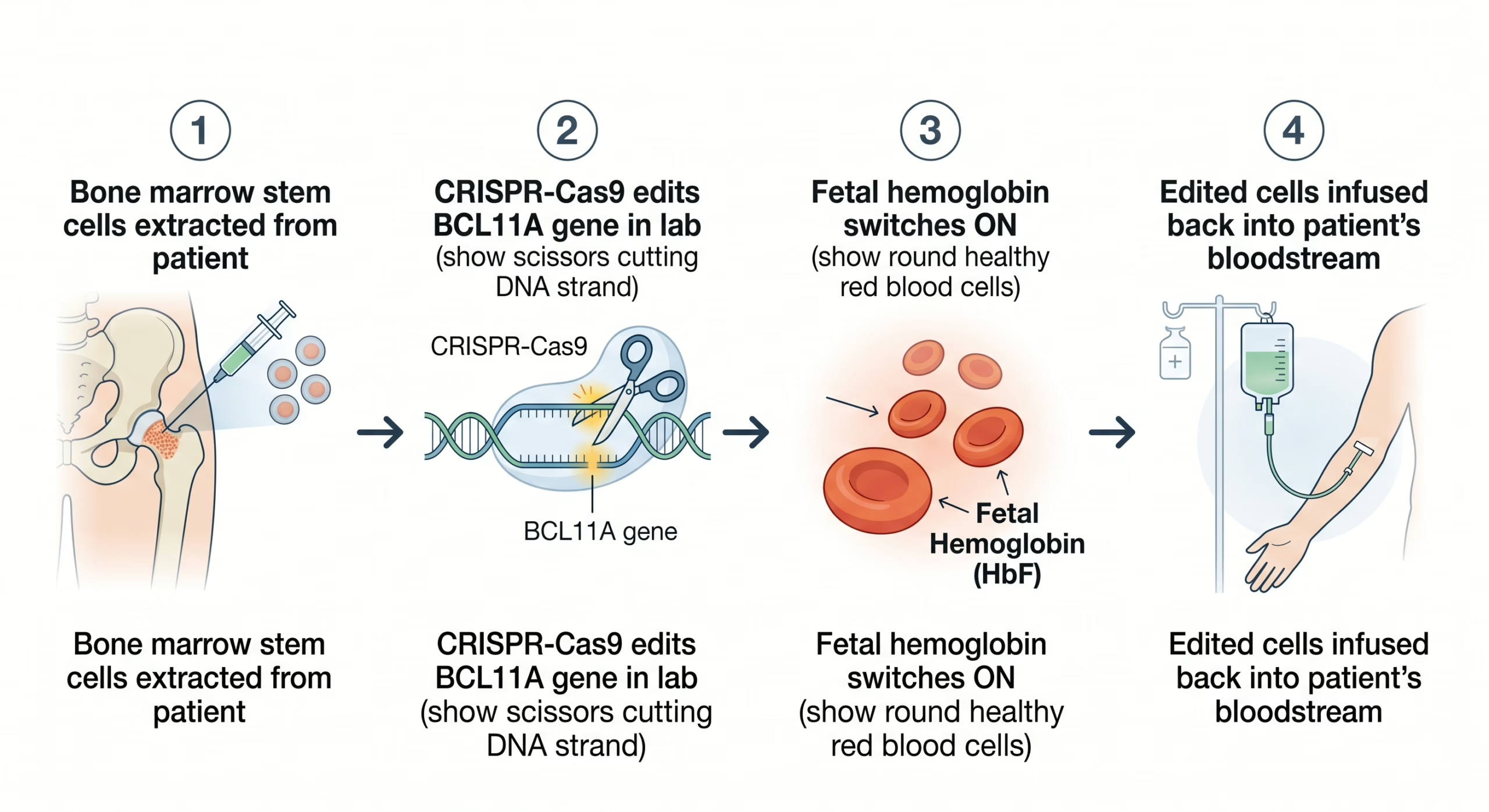
Casgevy does not directly fix the broken HBB gene. Instead, it uses a clever biological workaround rooted in our own developmental history.
Doctors collect hematopoietic stem cells (blood-forming stem cells) from the patient’s own bone marrow. These are the cells that produce all red blood cells for life.
In a lab, the CRISPR machinery is delivered into the stem cells. It targets and disrupts the BCL11A gene — a gene that normally silences fetal hemoglobin production after birth.
By knocking out BCL11A, the cells “forget” that they should stop making fetal hemoglobin (HbF). Fetal hemoglobin is naturally resistant to sickling — it was doing the job just fine before the body switched it off after birth.
After chemotherapy clears the existing bone marrow, the edited stem cells are returned to the patient’s bloodstream. They settle into the bone marrow and begin producing healthy, non-sickling red blood cells — for life.
The elegance of the approach is that it bypasses the broken gene entirely. Rather than correcting the mutation, it reactivates a backup system the body already has — one that worked perfectly before we were born.
— Key insight in the Casgevy mechanismWhat Do the Results Actually Show?
In clinical trials, the results were remarkable. Of the 29 patients treated with Casgevy and followed for at least 12 months, 28 — more than 96% — were completely free of severe vaso-occlusive pain crises for at least a year after treatment. Many showed a dramatic increase in fetal hemoglobin levels, often exceeding 40% of their total hemoglobin (normal adults have less than 1%).
A second CRISPR therapy approved the same day — Lyfgenia, made by bluebird bio — takes a different approach, directly inserting a functional copy of the hemoglobin gene. Both therapies represent a one-time, potentially permanent treatment.
The Bigger Picture: What This Means for Biology
Sickle cell disease is just the beginning. CRISPR-based therapies are now in clinical trials for dozens of conditions, including beta-thalassemia, certain forms of blindness, HIV, and multiple cancers. The FDA approval of Casgevy signals that the regulatory and safety framework for these therapies is maturing.
There are still significant challenges. The current treatment process is grueling and expensive — Casgevy is priced at approximately $2.2 million per patient. It also requires destroying existing bone marrow with chemotherapy before the edited cells are infused, which carries its own risks. Access remains deeply unequal.
But the biological proof-of-concept is now undeniable. We can take a cell out of a human body, rewrite a specific instruction in its DNA with molecular-level precision, put the cell back, and have it function correctly — possibly for the rest of that person’s life. That is one of the most profound demonstrations of applied biology in history.
- Sickle cell disease is caused by a single-letter mutation in the HBB gene, causing red blood cells to deform and clog blood vessels.
- CRISPR-Cas9 uses a guide RNA to find a precise DNA location and a Cas9 enzyme to cut it — acting as programmable molecular scissors.
- Casgevy doesn’t fix the broken gene directly — it reactivates fetal hemoglobin, a natural backup that doesn’t sickle.
- Over 96% of trial patients were free of severe pain crises after one treatment.
- This FDA approval is the first of its kind and opens the door to CRISPR therapies for dozens of other genetic diseases.
